Stablepharma awarded the highly competitive and prestigious EIC Accelorator Grant of €2.5mStablepharma Receives MHRA Approval for Phase 1 Trial of Fridge-Free Tetanus and Diphtheria (Td) Vaccine
The Reality
One of the biggest barriers to ensuring equitable access is the inability to deliver temperature sensitive pharmaceutical products, such as vaccines, globally while maintaining the cold chain. The high rate of wastage experienced during distribution and storage further exacerbate the issue. According to the World Health Organization, over 50% of vaccines are wasted annually, primarily due to failures in the logistical cold chain.
Fridge-Free Vaccines
Our novel StablevaX™ technology is designed to dramatically enhance the performance of existing and new vaccines, thermally stabilise and deliver a wide range of vaccines without the requirement for refrigeration.
Expanding Access
Stablepharma’s fridge-free vaccines can help expand access to patients and increase the success of immunisation programmes.
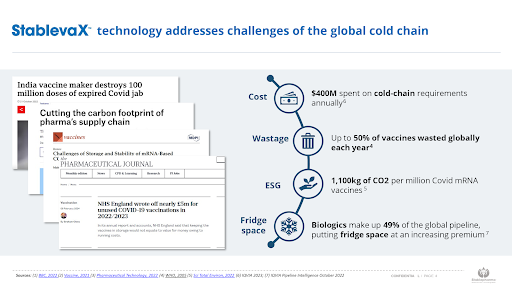
Stablepharma is pioneering a new era in pharmaceutical technology aimed at tackling wastage and reducing reliance on the global cold chain. Link to Technology Page Our mission is to:
- Improve global vaccine access
- Reduce vaccine wastage
- Support large-scale immunisation efforts
- Improve pandemic preparedness
- Lower the environmental footprint of the cold chain





