Category: Uncategorized


Stablepharma and AFT Pharmaceuticals partner to address multi-billion dollar anti-infective and oncology market
London, UK – 15 January 2026 – Stablepharma Limited, specialists in thermostable formulations, today announced a development and option-to-license agreement with AFT Pharmaceuticals to develop a portfolio of fridge-free sterile injectable medicines, initially targeting anti-infective and oncology treatments in a total global market valued at over USD 6 billion. Stablepharma will apply its proprietary stabilisation […]
Read More
Stablepharma Announces Database Lock for Fridge-Free Tetanus-Diphtheria Vaccine Clinical Trial
London, UK – 03 November 2025 – Stablepharma announced the database lock of its Phase 1 clinical trial of the world’s first, fridge-free, tetanus-diphtheria vaccine, SPVX02, which began recruitment in April this year. The trial evaluated the safety, tolerability and immunogenicity of SPVX02 against 2 comparator vaccines and enrolled 60 healthy participants at 2 UK […]
Read More
Revolutionising global vaccine access: The case for Stablepharma’s fridge-free vaccine technology
click here to read full article
Read More
Stablepharma enters Phase 1 Clinical Trials with world-first Fridge-Free Td Vaccine
Following MHRA approval, Stablepharma Ltd has commenced a Phase 1 clinical trial with its lead candidate, fridge-free tetanus and diphtheria vaccine, SPVX02. The Phase 1, first-in-human clinical trial led by Professor Saul Faust, is being conducted at the National Institute for Health and Care Research (NIHR) at Southampton Clinical Research Facility. Stablepharma’s pioneering science has […]
Read More
BIA Spotlight: how Stablepharma is pioneering the world’s first fridge-free Td vaccine
Following a number of ‘firsts’ for Stablepharma during 2024, we spoke with Ozgur Tuncer, CEO & Executive Director about a year that has accelerated the commercialisation of the world’s first fridge-free vaccine, SPVX02, for the prevention of Tetanus and Diphtheria. Ozgur also highlights some of the global challenges associated with the cold chain and talks […]
Read More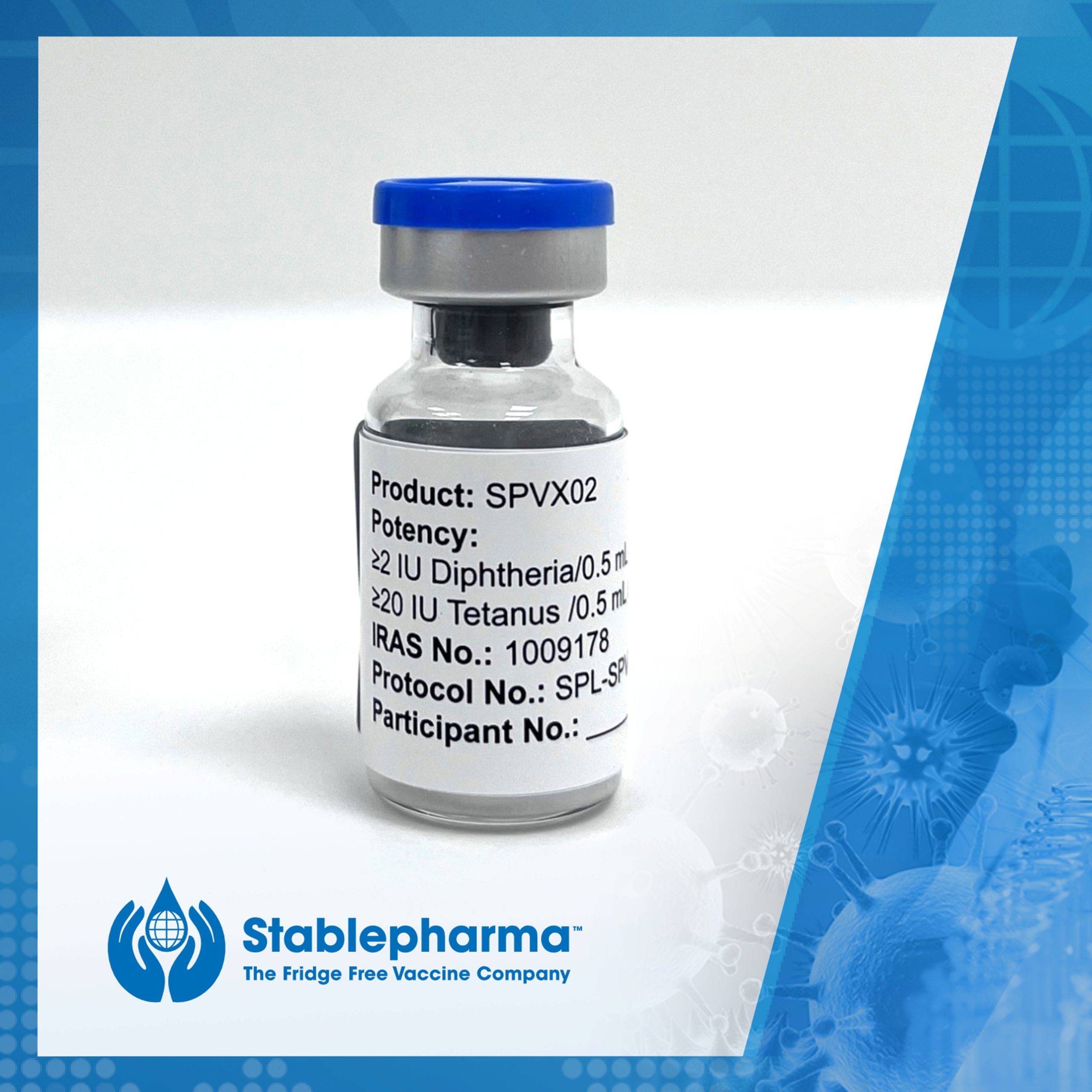
Stablepharma Receives MHRA Approval for Phase 1 Trial of Fridge-Free Tetanus & Diphtheria (Td) Vaccine
Stablepharma’s Fridge-Free Vaccine that Eliminates the Need for Cold Chain entering First-in-Human UK Clinical Trial. The UK’s Medicines and Healthcare products Regulatory Agency (MHRA) has granted approval for Stablepharma to begin a Phase I clinical trial with SPVX02, an adult booster vaccine aimed at preventing tetanus and diphtheria (Td) infections. This approval marks a major […]
Read More
Dr Kenny Simmen named Chief Business Officer of Stablepharma Ltd
Stablepharma has announced the appointment of Dr. Kenny Simmen as its Chief Business Officer, effective March 1, 2025. Following a year of service on the company’s Advisory Board, Dr. Simmen will now lead Stablepharma’s business development efforts, including partnering and licensing activities, as part of the company’s ambitious growth strategy. Dr Simmen has 30 years […]
Read More
Stablepharma Secures €2.5m EIC Grant, recognised Among Europe’s Top 71 Innovators
London, 19th February 2025 – UK-based biotechnology firm Stablepharma Ltd has been awarded €2.5 million through the prestigious European Innovation Council (EIC) Accelerator program. The grant recognises the company’s cutting-edge fridge-free pharmaceutical technology, which meets EIC’s rigorous criteria for excellence, impact, and risk management. Stablepharma is among 71 companies from 16 countries across Europe selected […]
Read More
Stablepharma named a Fierce Innovation Awards Winner!
Bath, United Kingdom, December 4th, 2024 – Stablepharma Ltd today announces it was named a Fierce Life Sciences Innovation Awards winner by Fierce Biotech and Fierce Pharma in the Biotech Innovation category. The awards program highlights and showcases outstanding innovation that is driving improvements and transforming the industry. “The Innovation Awards winners showcase the organizations that have […]
Read More